Answer:
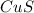
Step-by-step explanation:
A double displacement reaction is one in which exchange of ions take place. The salts which are soluble in water are designated by symbol (aq) and those which are insoluble in water and remain in solid form are represented by (s) after their chemical formulas.
Precipitation is a double displacement reaction in which one of the product is formed in solid state called as precipitate.
The balanced chemical reaction for ammonium sulphide and copper (II) nitrate is:
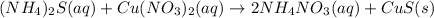
Thus
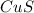 is formed as a precipitate.
is formed as a precipitate.