Answer:
9.03 × 10²² oxygen molecules.
Step-by-step explanation:
We want to determine the number of oxygen molecules in 3.36 L of oxygen gas at STP.
Recall that at STP, for any gas, it has a volume of 22.4 liters per mole.
Additionally, recall that there are 6.02 × 10²³ substances in one mole of a substance.
With the given value, hence multiply:
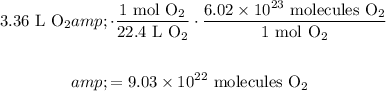
In conclusion, there are aobut 9.03 × 10²² oxygen molecules in 3.36 L of oxygen as at STP.