Step-by-step explanation:
According to mole concept
a.
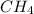 Moles to
Moles to
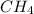 grams
grams
Number of moles =
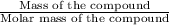
Mass of
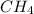 = Moles of
= Moles of
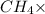 16 g/mol
16 g/mol
b.
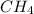 Liters to
Liters to
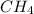 moles at STP.
moles at STP.
At STP,1 mol gas occupies 22.4 L of volume.
Multiply the given volume of gas in L with
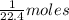
c.Molecules
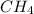 of to moles
of to moles
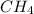
1 mole =
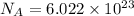 atoms or molecules
atoms or molecules
So, in 1 molecule =
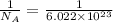 mole.
mole.
Multiply the given number of molecules with
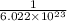 mole to get the number of moles of
mole to get the number of moles of
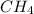 .
.