Step-by-step explanation:
Molarity is defined as the number of moles in liter of solution.
Mathematically, Molarity =
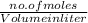
The data is given as volume is 1.6 liters and molarity is 0.75 M. Therefore, calculate the number of moles as follows.
Molarity =
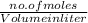
or, No. of moles =
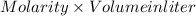
= 0.75 M × 1.6 liter
= 1.2 moles
Thus, we can conclude that there are 1.2 moles of glucose in 1.6 liters of a 0.75 M C6H12O6 solution.