Answer : The number of moles of air present is, 0.055 mole
Solution :
Using ideal gas equation :
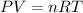
where,
P = pressure of the gas = 750 torr = 0.98 atm (1 atm = 760 torr)
V = volume of the gas = 1.35 L
T = temperature of the gas =
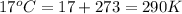
R = gas constant = 0.0821 Latm/moleK
n = number of moles of air = ?
Now put all the given values in this formula, we get the number of moles of air present.
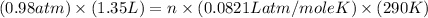
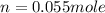
Therefore, the number of moles of air present is, 0.055 mole