Answer:
False.
Step-by-step explanation:
Hello,
In this case, as the moles are held constant, one uses the Gay-Lussac's law in order to relate both the temperature and the pressure at the beginning and at the end of the experiment via the following relationship:
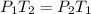
By solving for the pressure at the second (final) state we can substantiate whether the 1.5 atm are correct as shown below:
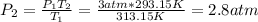
Therefore, even when the pressure is correctly decreased, the given value is wrong as 2.8 atm is the correct final pressure, in this manner, the statement is false.
Best regards.