Answer : The number of moles of hydrogen gas needed are, 0.253 mole
Explanation : Given,
Moles of water = 0.253 mole
Now we have to calculate the moles of hydrogen gas.
The given balanced chemical reaction is,
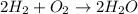
From the balanced reaction we conclude that,
As, 2 moles of water obtained from 2 moles of hydrogen gas
So, 0.253 moles of water obtained from
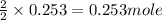 of hydrogen gas
of hydrogen gas
Therefore, the number of moles of hydrogen gas needed are, 0.253 mole