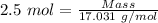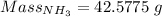Answer:
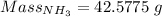
Step-by-step explanation:
Moles of
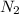 :-
:-
Mass = 35.00 g
Molar mass of
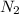 = 28.014 g/mol
= 28.014 g/mol
The formula for the calculation of moles is shown below:
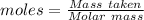
Thus,
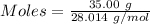
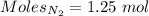
According to the reaction:-
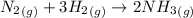
1 mole of
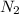 on reaction forms 2 moles of
on reaction forms 2 moles of
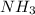
1.25 mole of
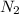 on reaction forms 2*1.25 moles of
on reaction forms 2*1.25 moles of
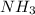
Moles of
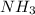 = 2.5 moles
= 2.5 moles
Molar mass of
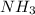 = 17.031 g/mol
= 17.031 g/mol
The formula for the calculation of moles is shown below:
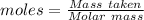
Thus,