Answer: The charge on the lithium atom when it looses one electron is +1
Step-by-step explanation:
An ion is formed when a neutral atom looses or gains electrons.
- When an atom looses electrons, it results in the formation of positive ion known as cation.
- When an atom gains electrons, it results in the formation of negative ion known as anion.
We are given:
A chemical element of lithium having atomic number '3'
The electronic configuration of this element is
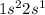
If, this element looses 1 electron, now the number of electrons left is 2
Number of protons = 3
Number of electrons = 2
Charge = Number of protons - Number of electrons = 3 - 2 = +1
Hence, the charge on the lithium atom when it looses one electron is +1