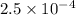Answer: The hydrogen ion concentration for the given pH value is
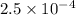
Step-by-step explanation:
pH is defined as the negative logarithm of hydrogen ion or hydronium ion concentration. The equation used to represent pH of the solution follows:
![pH=-\log[H^+]](https://img.qammunity.org/2018/formulas/chemistry/high-school/9so9u3mmeurzbcqkpf3jt5b89ctmc03tdp.png)
We are given:
pH of the solution = 3.6
Putting values in above equation, we get:
![3.6=-\log[H^+]](https://img.qammunity.org/2018/formulas/chemistry/high-school/taqca3fygkluw5zz6znylx3iu8okh45njx.png)
![[H^+]=2.5* 10^(-4)](https://img.qammunity.org/2018/formulas/chemistry/high-school/tsujxal54pe5swninl02oowlgia9ns8kqw.png)
Hence, the hydrogen ion concentration for the given pH value is