Answer: The correct answer is Option 1.
Step-by-step explanation:
1-pentyne is a hydrocarbon which has a triple bond present between carbon and carbon atoms. This is considered as an unsaturated hydrocarbon and an alkyne.
To form a single bond, number of electrons required are 2.
Here, triple bond is present between carbon and carbon atoms, so number of electrons required to form this bond will be
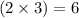
Hence, the correct answer is Option 1.