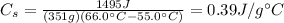The amount of heat needed to increase the temperature of a substance by

is given by
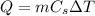
where m is the mass of the substance, Cs is its specific heat capacity and

is the increase of temperature.
If we re-arrange the formula, we get
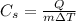
And if we plug the data of the problem into the equation, we can find the specific heat capacity of the substance: