Answer:
there are 1.49 grams of glucose in 100 mL of the final solution
Step-by-step explanation:
First, calculate the initial concentration of glucose. As the concentration is the rate between amount of solute (glucose) and solution, we can express it as grams of glucose per volume of solution:
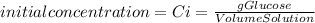
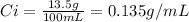
As we are diluting the solution, we use the equation:
(Initial concentration)(initial volume)=(final concentration)(final volume) or (Ci)(Vi)=(Cf)(Vf)
We need the final concentration of glucose to find the grams of glucose in 100 mL of this solution, from the above equation we have:
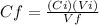
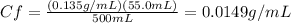
Note that 0.500 L = 500 mL, so the units cancelled each other.
And for the definition of concentration we have:
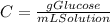
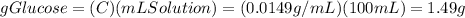
So, there are 1.49 grams of glucose in 100 mL of the final solution which concentration is 0.0149 g/mL