Data Given: Molarity = 0.085 M
Ka = 4.5 × 10⁻⁴
To Find: pH = ?
Solution: First find out the concentration of [H⁺] Ions using following formula,
[H⁺] =
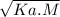
Puting values,
[H⁺] =
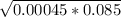
[H⁺] =
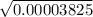
[H⁺] =
0.00618465843
Or,
[H⁺] = 6.18 × 10⁻³
Calculate pH as,
pH = -log [H⁺]
pH = -log (6.18 × 10⁻³)
pH = 2.20