Answer: The pH of the solution is 0.125
Step-by-step explanation:
pH is the negative logarithm of hydrogen ion concentration present in a solution.
To calculate the pH of the reaction, we use the equation:
![pH=-\log[H^+]](https://img.qammunity.org/2019/formulas/chemistry/high-school/vwilut25e4cux34589pwoorivy6w6y51xe.png)
The chemical equation for the dissociation of nitric acid follows:
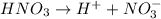
By stoichiometry of the reaction:
1 mole of nitric acid produces 1 mole of hydrogen ions and 1 mole of nitrate ions.
So,
![[H^+]=0.75M](https://img.qammunity.org/2019/formulas/chemistry/college/b6kwlr8u7mqevmvsgh68kst0houcuarh43.png)
Putting values in above equation, we get:
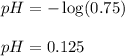
Hence, the pH of the solution is 0.125