Answer:
b. Endothermic
Step-by-step explanation:
Hello,
By considering the equilibrium chemical reaction:
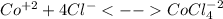
The Le Chatelier's principle states that if the temperature for endothermic chemical reactions is increased the equilibrium will shift rightwards, in such a way, as at high temperatures the blue color, corresponding to the complex, predominates, this behavior assures the chemical reaction as endothermic.
Best regards.