Charles law gives the relationship between volume and temperature of gas.
it states that at constant pressure volume of gas is directly proportional to temperature.
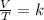
where V - volume
T - temperature in kelvin
and k - constant
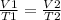
where parameters for the first instance are on the left side and parameters for the second instance are on the right side of the equation
T1 - 25 °C + 273 = 298 K
T2 - 35 °C + 273 = 308 K
substituting the values in the equation
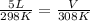
V = 5.17 L
new volume is 5.17 L