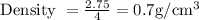The density of the material is
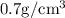
Answer: Option D
Step-by-step explanation:
Density of any material is defined as the amount of mass present in a specified volume. So density is directly proportional to the mass of the object and inversely proportional to the volume in which the mass is present.
So the density will be calculated as the ratio of mass to the volume. Thus as the mass of the unknown materials is given as 2.75 g present in a volume of
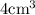 , the density will be
, the density will be
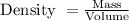
Thus,