Answer:
The volume of the balloon is 4.92 L at the height of 4 km.
Step-by-step explanation:
Initial Volume of the helium gas
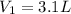
Initial pressure of the helium gas
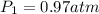
Final volume of the helium gas =
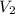
Final pressure of the helium gas =
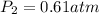
Applying Boyle's law:
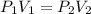 (At constant temperature)
(At constant temperature)
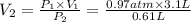
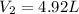
The volume of the balloon is 4.92 L at the height of 4 km.