Answer: D)
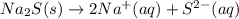
Step-by-step explanation: Decomposition reaction is a chemical reaction in which one reactant decomposes to give two or more than two products.
The substances which are soluble in water are designated by symbol (aq) and those which are insoluble in water and remain in solid form are represented by (s) after their chemical formulas.
Thus when sodium sulfide dissociates in aqueous solution, it gives ions which are present in aqueous form.