Answer:
The water pressure at 105 ∘ C is 908.7641 mmHg.
Step-by-step explanation:
The Antoine equation describes the relationship between the temperature and the vapor pressure of pure substances. In other words, it is an equation used to obtain the value of the vapor pressure as a function of temperature for pure components, and is based on a correlation of three parameters A, B and C. The Antoine equation is:
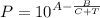
where:
- P is the vapor pressure of water, usually in mmHg.
- T is the water temperature in celsius.
- A, B and C are the Antoine constants for water. The values of A, B and C depend on the units used for the vapor pressure and the temperature and the temperature range at which the correlation was obtained.
In the image you can see the values of the Antoine parameters for two temperature ranges for water. In these data the unit of temperature is ° C and the pressure to be calculated will be in mmHg.
In this case, since the temperature is 105 ° C, the temperature range of interest from 99 to 374 ° C. Then the parameters will have the following values: A = 8.14019, B = 1810.94 and C = .244.485
Replacing in the previous equation you get:
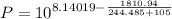
Solving, you get as a result:
P=908.7641 mmHg
The water pressure at 105 ∘ C is 908.7641 mmHg.