The dissociation of Ca(OH)₂ in aqueous medium is given below:
Ca(OH)₂(aq) ⇄ Ca⁺²(aq) + 2 OH⁻
The molarity of Ca(OH)₂ =
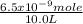
= 6.5 x 10⁻¹⁰ M Ca(OH)₂
The solubility product (Ksp) of Ca(OH)₂ = 6.5 x 10⁻⁶
The solubility (S) of Ca(OH)₂ is :
Ksp = [Ca⁺²][OH⁻]² = S (2S)² = 4 S³
6.5 x 10⁻⁶ = 4 S³
S = 0.0117 M
[OH⁻] = 2 S = 2 * (0.0117 M) = 0.0234 M
Each mole of Ca(OH)₂ gives two moles of OH⁻ ions therefore;
[OH⁻] = 2 * (6.5 x 10⁻¹⁰ M) = 1.3 x 10⁻⁹ M
So the concentration of [OH⁻] ions less then the saturated [OH⁻] conc.
⇒ The solution is not precipitated
Since [OH⁻] < 10⁻⁷
Total conc. [OH⁻] = (10⁻⁷ due to ionization of water + 1.3 x 10⁻⁹ M)
= 1.013 x 10⁻⁷ M
pOH = - log (1.013 x 10⁻⁷ M) = 6.957
pH = 14 - pOH = 7.043