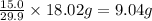Answer:
9.04 g of
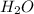 are needed to react
are needed to react
Step-by-step explanation:
According to balanced equation, 1 mol of
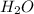 reacts with 1 mol of
reacts with 1 mol of
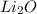
Molar mass of
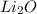 = 29.9 g/mol
= 29.9 g/mol
Number of moles = (mass)/(molar mass)
So, 15.0 g of
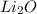 =
=
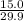 moles of
moles of
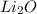
So, moles of
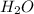 are needed to react =
are needed to react =
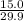 moles
moles
Molar mass of
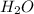 = 18.02 g/mol
= 18.02 g/mol
So, mass of
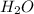 are needed to react =
are needed to react =