We are given following information:
n=0.650mol
T=30°C
V=0.700L
First step is to transform these numbers into basic units:
n=0.650mol
T=30°C = 303.15K
V=0.700L = 0.0007m³
We are looking for pressure p.
The formula that connects all these variables is Ideal gas formula:
p * V = n * R * T
where R is gas constant.
We solve this formula for p:
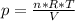
Now we insert values and calculate the pressure.
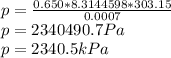
Correct answer is A).
B) is not correct because 1atm = 101325Pa so the value is p=2340490.7 / 101325 = 23.1 atm.