Answer:There are 4.32 moles of Cr in 2.16 moles of
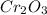 .
.
Step-by-step explanation:
Moles of
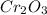 = 2moles
= 2moles
In one molecule of
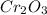 there are 2 atoms of Cr.
there are 2 atoms of Cr.
Then in 1 Mole of
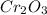 contains = 2 moles of Cr
contains = 2 moles of Cr
Number of moles of Cr in 2.16 moles of
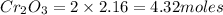 of Cr.
of Cr.
There are 4.32 moles of Cr in 2.16 moles of
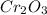 .
.