The chemical reaction in which one component of each reactant molecules gets exchanged to form products are known as a double-displacement reaction.
The given double-displacement reaction with there respective phases is:
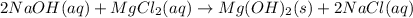
Precipitation of
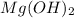 takes place and the
takes place and the
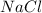 will remain in the solution in aqueous phase.
will remain in the solution in aqueous phase.
Thus, option b is correct.