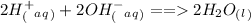The salt produced is
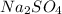 . The reaction between a strong acid and a strong base results in salt and water. Since the base and acid are strong the reaction goes to completion. The balanced equation is:
. The reaction between a strong acid and a strong base results in salt and water. Since the base and acid are strong the reaction goes to completion. The balanced equation is:
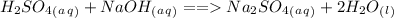
In fact this is a neutralization reaction in which
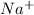 and
and
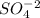 are spectator ions.
are spectator ions.
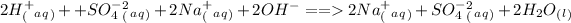 ,
,
If you remove the spectactor ions,
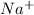 ions and
ions and
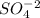 on both sides of the equation, then the net ionic equation is,
on both sides of the equation, then the net ionic equation is,