Answer : The volume of neon gas will be, 7.99 liters.
Explanation : Given,
Mass of argon (Ar) gas = 27.1 g
Molar mass of argon = 39.95 g/mole
Volume of argon gas = 4.21 L
Moles of neon (Ne) gas = 1.29 mole
First we have to calculate the moles of argon gas.
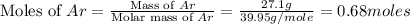
Now we have to calculate the volume of neon gas.
According to the Avogadro's law, the volume of gas is directly proportional to the number of moles of gas at same pressure and temperature. That means,
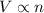
or,
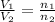
where,
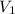 = volume of argon gas
= volume of argon gas
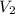 = volume of neon gas
= volume of neon gas
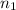 = number of moles of argon gas
= number of moles of argon gas
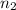 = number of moles of neon gas
= number of moles of neon gas
Now we put all the given values in this formula, we get
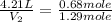
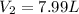
Therefore, the volume of neon gas will be, 7.99 liters.