Answer:
1.03mole
Explanation:
Given parameters:
Pressure = 0.884 atm
Temperature = 6°C = 273 + 6 = 279k
Volume = 26.5L
Unknown:
Number of moles = ?
Solution:
To solve this problem, we use the ideal gas equation:
PV = nRT
P is the pressure
V is the volume
n is the number of moles
R is the gas constant = 0.082atmdm³mol⁻¹K⁻¹
T is the temperature
n =
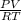 =
=
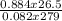 = 1.03mole
= 1.03mole