Answer:
The percent of CaCO₃ in the sample is 75%.
Step-by-step explanation:
We can find the percent of CaCO₃ in the sample knowing that 30 g of Ca are present in 100 g of CaCO₃.
First, we need to find the number of moles of Ca, and since the ratio of Ca in CaCO₃ is 1:1 we can find the percent of CaCO₃ in the sample as follows:
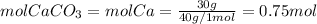
Now, the mass of CaCO₃ is:
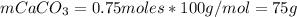
Finally, the percent of CaCO₃ in 100 g of the sample is:
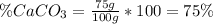
Therefore, the percent of CaCO₃ in the sample is 75%.
I hope it helps you!