Answer : The Lewis-dot structure of
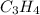 is shown below.
is shown below.
The number of sigma and pi-bonds are 6 and 2 respectively.
Explanation :
Lewis-dot structure : It shows the bonding between the atoms of a molecule and it also shows the unpaired electrons present in the molecule.
In the Lewis-dot structure the valance electrons are shown by 'dot'.
The given molecule is,
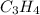
As we know that carbon has '4' valence electrons and hydrogen has '1' valence electron.
Therefore, the total number of valence electrons in
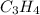 = 3(4) + 4(1) = 16
= 3(4) + 4(1) = 16
According to Lewis-dot structure, there are 16 number of bonding electrons and 0 number of non-bonding electrons.
The number of sigma and pi-bonds are 6 and 2 respectively.