Answer:
See explanation.
Step-by-step explanation:
Hello!
In this case, since we know the heat of reaction per gram of reactant and we should know the total energy of reaction, but it is not there, we are going to assume it is 1200 J as usual in these problems, so you can change it to whatever your given heat is.
In such a way, we set up the math as shown below:
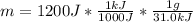
Which results:

Best regards!