Answer:
Yes
Step-by-step explanation:
m = Mass of glycerin = 168 g
Volume of glycerin that can fit in the container = 150 mL
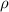 = Density of glycerin = 1.26 g/mL
= Density of glycerin = 1.26 g/mL
Volume the given mass of glycerin will occupy is
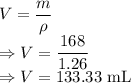
A mass of 168 g of glycerin will have a volume of 133.33 mL which is less than the 150 mL flask. So, yes the flask is large enough to fit 168 g of glycerin.