Answer: 20.0 g of hydrogen chloride must simultaneously be formed
Step-by-step explanation:
The balanced chemical reaction is :
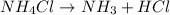
According to the law of conservation of mass, mass can neither be created nor be destroyed. The mass on reactant side must be equal to the mass on product side.
Thus mass of reactants = mass of products
Given : mass of ammonium chloride = mass of reactants = 29.4 g
mass of ammonia = 9.4 g
mass of products = mass of ammonia + mass of hydrogen chloride
9.4 g +mass of hydrogen chloride = 29.4 g
mass of hydrogen chloride = 20.0 g