This reaction would give rise to two products.
- 2-bromo-3-methylhexane, and
- 3-bromo-3-methylhexane.
However, 2-bromo-3-methylhexane would be more common than 3-bromo-3-methylhexane among the products.
The hydrogen atom in a hydrogen bromide molecule carries a partial positive charge. It is attracted to the double bond region with a high electron density. The hydrogen-bromine bond breaks when HBr gets too close to a double bond to produces a proton
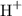 and a bromide ion
and a bromide ion
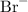 .
.
The proton would attack the double bond to produce a carbocation. It could attach itself to either the second or the third carbon atom.
Carbocations are unstable and might decompose over time. The first carbocation is more stable than the second for having three alkyl groups- i.e., straight carbon chains- attached to the center of the positive charge. Alkyl groups have stabilizing positive induction effect on positively-charged carbon. The second carbocation has only two, and is therefore not as stable. The first carbocation thus has the greatest chance to react with a bromide ion to produce a stable halocarbon.
Bromide ions are negatively charged. They attach themselves to carbocations at the center of positive charge. Adding a bromide ion to the first carbocation would produce 3-bromo-3-methylhexane whereas adding to the second produces 2-bromo-3-methylhexane.
The most likely product of this reaction is therefore 3-bromo-3-methylhexane.