Answer: Option (c) is the correct answer.
Step-by-step explanation:
A synthesis reaction is defined as a reaction in which two substances or atoms combine together to result in the formation of a compound.
For example,
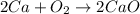
Here, calcium and oxygen combine together to form compound calcium oxide.
Whereas in a decomposition reaction, a compound decomposes into two atoms.
A single displacement reaction is a reaction in which one element is displaced by another element.
For example,
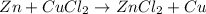
A double displacement reaction is a reaction in which elements of two compounds interchange with each other forming two new compounds.
For example,
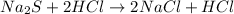
Hence, we can conclude that given reaction is a synthesis reaction.