Answer:- 475 mL.
Solution:- It's based on Boyle's gas law. At constant temperature, "The volume of the gas is inversely proportional to the pressure."
Equation for this law is written as:
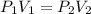
P and V stands for pressure and volume where as the subscripts 1 and 2 are for initial and final values.
Initial pressure is 6.20 atm and it asks to calculate the initial volume. Final pressure is 9.150 atm and the final volume is 322 mL.
Let's plug in the values in the equation and solve it for initial volume:
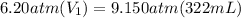
On dividing both sides by 6.20 atm:
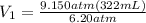
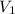 = 475 mL
= 475 mL
So, the originally the gas container volume was 475 mL.