Answer : Half life and radioactive decay are inversely proportional to each other.
Explanation :
The mathematic relationship between the half-life and radioactive decay :
 ................(1)
................(1)
where,
N = number of radioactive atoms at time, t
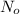 = number of radioactive atoms at the beginning when time is zero
= number of radioactive atoms at the beginning when time is zero
e = Euler's constant = 2.17828
t = time
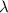 = decay rate
= decay rate
when
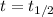 then the number of radioactive decay become half of the initial decay atom i.e
then the number of radioactive decay become half of the initial decay atom i.e
 .
.
Now substituting these conditions in above equation (1), we get
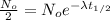
By rearranging the terms, we get
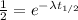
Now taking natural log on both side,
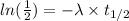
By rearranging the terms, we get
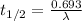
This is the relationship between the half-life and radioactive decay.
Hence, from this we conclude that the Half life and radioactive decay are inversely proportional to each other. That means faster the decay, shorter the half-life.