as per the reaction we have

so here 1 mole of H2 react with 1 mole of O2 and give 1 mole of H2O2 as the product
now we will find the moles of given reactant
moles of H2
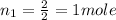
moles of O2
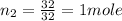
so here both present in 1 mole each hence the product will also formed 1 mole
so here we have
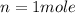 H2O2 will form
H2O2 will form
so mass of the product
mass = moles* molecular mass
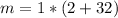
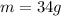
so the product mass will be 34 g