Answer : The number of moles of water formed can be 116 moles.
Explanation :
First we have to calculate the limiting and excess reagent.
The balanced chemical reaction will be:
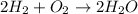
From the balanced chemical reaction we conclude that,
As, 1 mole of
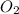 react with 2 mole of
react with 2 mole of
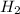
So, 58 moles of
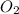 react with
react with
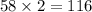 moles of
moles of
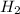
From this we conclude that,
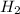 is an excess reagent because the given moles are greater than the required moles and
is an excess reagent because the given moles are greater than the required moles and
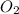 is a limiting reagent and it limits the formation of product.
is a limiting reagent and it limits the formation of product.
Now we have to calculate the moles of
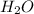
From the reaction, we conclude that
As, 1 mole of
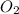 react to give 2 mole of
react to give 2 mole of
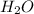
So, 58 moles of
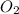 react to give
react to give
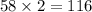 moles of
moles of
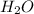
Therefore, the number of moles of water formed can be 116 moles.