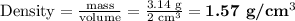The density of the mixture will be 1.57 g/cm³.
Step 1. Calculate the mass of the butter.
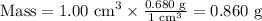
Step 2. Calculate the mass of the sand.
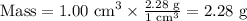
Step 3. Calculate the density of the mixture.
Total mass = 0.860 g + 2.28 g = 3.14 g.
Total volume = 1 cm³ + 1 cm³ = 2 cm³