You must use 134 g O₂ to produce 118 g H₂O.
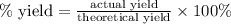
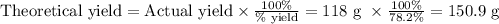
M_r: 32.00 18.02
2H₂ + O₂ ⟶ 2H₂O
Moles of H₂O = 150.9 g H₂O × (1 mol H₂O/18.02 g H₂O) = 8.374 mol H₂O
Moles of O₂ = 8.374 mol H₂O × (1 mol O₂/2 mol H₂O) = 4.187 mol O₂
Mass of O₂ = 4.1877 mol O₂ × (32.00 g O₂/1 mol O₂) = 134 g O₂