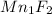Answer : The empirical formula of manganese fluoride is
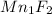 .
.
Solution : Given,
If percentage are given then we are taking total mass is 100 grams.
So, the mass of each element is equal to the percentage given.
Mass of Mn = 59.1 g
Mass of F = 40.9 g
Molar mass of Mn = 54.94 g/mole
Molar mass of F = 18.99 g/mole
Step 1 : convert given masses into moles.
Moles of Mn =
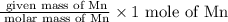 =
=
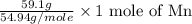 = 1.076 moles
= 1.076 moles
Moles of F =
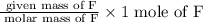 =
=
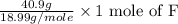 = 2.154 moles
= 2.154 moles
Step 2 : For the mole ratio, divide each value of moles by the smallest number of moles calculated.
For Mn = 1.076/1.076 = 1
For F = 2.154/1.076 ≈ 2
Mn : F = 1 : 2
The mole ratio of the element is represented by subscripts in empirical formula.
Therefore, the Empirical formula =