Answer:
1. A) Atoms gain valence electrons to form anions.
2. D) 50.94151 amu
3. D) elements in clouds of gas and dust in deep space.
4. D) silicon
Step-by-step explanation:
Hello,
1. In this question, we consider that anions are chemical ions negatively charged, it means that they gain valence electrons to increase their oxidation states, thus the answer is: A) Atoms gain valence electrons to form anions.
2. In order to compute the required average atomic mass, we must consider the mass of each isotope and its percentage as shown below:
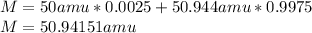
Therefore answer is D).
3. In this case, the answer is: D) elements in clouds of gas and dust in deep space, as each element has its own atomic emission spectrum, therefore one can identify the spectra in order to determine which elements are present.
4. Since helium is obtained via the atomic fusion of two hydrogen atoms, nickel can be obtained via the atomic fusion of two silicon atoms since each silicon has 14 protons and electrons, thus, if we fusion them we obtain 28 protons and electrons, validating the existence of a nickel atom.
Best regards.