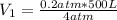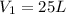Answer:
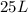
Step-by-step explanation:
The problem can be solved using Boyle's law for the volume and pressure of a gas at constant temperature:
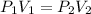
Where the quantities with sub-index 1 are the initial conditions for the gas and the quantities with sub-index 2 are for the final conditions of the gas.
We want to know the initial volume, this is:
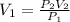
we also know from the problem that
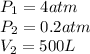
Thus, the initial volume V1 is: