Answer: The half life of the decomposition reaction is 0.204 s
Step-by-step explanation:
The equation used to calculate half life for first order kinetics:
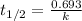
where,
k = rate constant of the reaction =
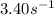
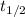 = half life of the reaction = ?
= half life of the reaction = ?
Putting values in above equation, we get:
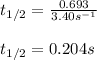
Hence, the half life of the decomposition reaction is 0.204 s