Answer:
Step-by-step explanation:
Hello,
In this case, starting by the ideal gas constant as shown below:
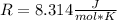
In order to get the requested value, the following conversion should be applied:
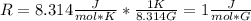
Therefore, the obtained temperature scale implies that:
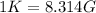
Best regards.