Answer: The mass of mercury (II) sulfide is 53.51 g
Step-by-step explanation:
To calculate the number of moles, we use the equation:
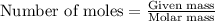 .....(1)
.....(1)
- For mercury (II) nitrate:
Given mass of mercury (II) nitrate = 126.27 g
Molar mass of mercury (II) nitrate = 324.7 g/mol
Putting values in equation 1, we get:
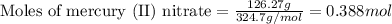
Given mass of sodium sulfide = 17.796 g
Molar mass of sodium sulfide = 78.04 g/mol
Putting values in equation 1, we get:
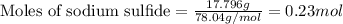
The chemical equation for the reaction of Mercury (II) nitrate and sodium sulfide follows:
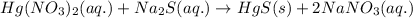
By Stoichiometry of the reaction:
1 mole of sodium sulfide reacts with 1 mole of mercury (II) nitrate
So, 0.23 moles of sodium sulfide will react with =
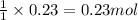 of mercury (II) nitrate
of mercury (II) nitrate
As, given amount of mercury (II) nitrate is more than the required amount. So, it is considered as an excess reagent.
Thus, sodium sulfide is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
1 mole of sodium sulfide produces 1 mole of mercury (II) sulfide
So, 0.23 moles of sodium sulfide will produce =
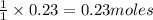 of mercury (II) sulfide
of mercury (II) sulfide
Now, calculating the mass of mercury (II) sulfide from equation 1, we get:
Molar mass of mercury (II) sulfide = 232.66 g/mol
Moles of mercury (II) sulfide = 0.23 moles
Putting values in equation 1, we get:
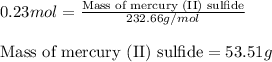
Hence, the mass of mercury (II) sulfide is 53.51 g