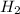Answer: B. A covalent bond in which the electrons are shared equally
Step-by-step explanation:
Covalent bonds are formed by sharing of electrons between non metals.
A polar covalent bond is defined as the bond which is formed when there is a difference of electronegativities between the atoms. It is also defined as the bond which is formed due to the unequal sharing of electrons between the atoms. Example:
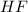
Non-polar covalent bond is defined as the bond which is formed when there is no difference of electronegativities between the atoms. It is also defined as the bond which is formed due to the equal sharing of electrons between the atoms. Example: