Radius of nuclei is given by formula
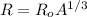
now we can say volume of the nuclei is given as
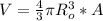
now the density is given as
density = mass / volume
mass of nuclei = mass of neutron + mass of protons
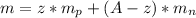
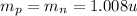

Now density is given as
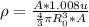
here we know that
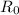 = 1.2 fm
= 1.2 fm
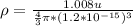
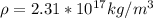
So from above we can say that density of all nuclei is almost same.